
基本信息
姓名:黄俊
学位:博士
职称:特聘教授
研究领域(方向):天然产物和小分子药物合成
联系方式:
邮箱:junhuang@usc.edu.cn, junhuang@pku.edu.cn
个人简介
黄俊,特聘教授、博士生导师。主要从事天然产物和小分子药物合成和工艺研究。近五年,以通讯作者(含共同)在国际著名期刊J. Am. Chem. Soc,Angew. Chem. Int. Ed,JACS Au等发表多篇学术论文。主持国家自然科学基金面上项目和青年项目,授权中国发明专利6项,国际专利1项。获湖南省湖湘青年英才资助。
教育和工作经历
2021/10 至今 伟德国际victor1946 特聘教授
2018/10-2021/10 北京大学 副研究员
2015/08-2016/09 纽约州立大学 博士后 (导师:Zhang Wang,现UC Davis)
2010/09-2015/07 北京大学 博士 (导师:杨震教授)
研究内容和特色
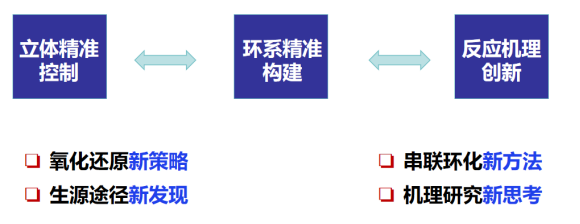
天然产物是有机小分子药物研发的重要物质来源。大多天然产物也面临着来源稀缺以及合成难度较大的主要问题。简洁、高效的天然产物全合成为具有重要生物活性和独特化学结构的天然产物后续成药性研究提供物质基础。发展丰富多样合成方法学和新颖高效合成策略,基于汇聚式和集群式合成理念,实现活性天然产物的简洁高效全合成研究为当前有机合成化学的研究热点之一。课题组立足复杂天然产物全合成中碳碳键立体选择性精准合成的核心科学难题,围绕氢原子/质子转移途径介导的碳碳键精准构筑和复杂环系立体选择性构建,发展多样性合成方法学和高效合成策略,实现天然产物和小分子药物的精准合成。
代表性成果及论文
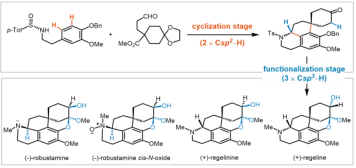
1. Liuyang Pu, Zhenbao Wang, JinYu Xia, Zhihong Chen, Miao Xiao, Lei Zhu, and Jun Huang*. Divergent Asymmetric Synthesis of Four Pentacyclic Homoproaporphine Alkaloids via C-H Elaboration. J. Am. Chem. Soc. 2026, 148, doi.org/10.1021/jacs.6c00213.
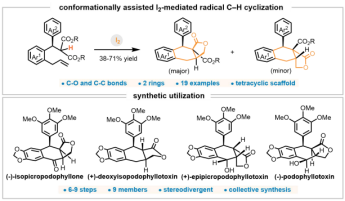
2. Rong Yin, Xudong Wang, Rui Zhou, Lei Zhu, and Jun Huang*. Unified Asymmetric Synthesis of Aryltetralin Lactone Cyclolignans via Conformation-Assisted Radical C−H Cyclization. JACS Au 2025, 5, 5690−5697.
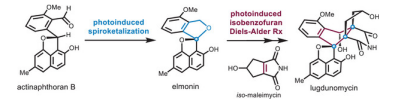
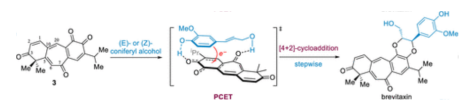
3. Lei Zhu, and Jun Huang*. Total Synthesis of Lugdunomycin via Sequential Photoinduced Spiroketalization and Isobenzofuran Diels-Alder Reactions. Angew. Chem. Inter. Ed. 2025, 64, e202422615.
4. Miao Xiao, Liuyang Pu, Qiaoli Shang, Lei Zhu, Jun Huang*. Asymmetric total synthesis of tricyclic prostaglandin D2 metabolite methyl ester via oxidative radical cyclization. Beilstein J. Org. Chem. 2025, 21, 1964-1972. (Invited, Guest Editor: Yefeng. Tang )
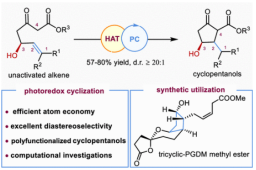
5. Miao Xiao#, Qiaoli Shang#, Liuyang Pu, Zheyuan Wang, Lei Zhu*, Zhen Yang*, and Jun Huang*. Photoredox-Catalyzed Radical Cyclization of Unactivated Alkene-Substituted β-Ketoesters Enabled Asymmetric Total Synthesis of Tricyclic Prostaglandin D2 Metabolite Methyl Ester. JACS Au. 2025, 5, 1367-1375.
6. Yang Lu#, Tingting Cao#, Kang Li, Ying-Wu Lin, Lei Zhu*, and Jun Huang*. Total Synthesis of Brevitaxin. Org. Lett. 2024, 26, 5237-5242.
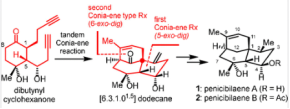
7. Zheyuan Wang, Zhilin Song, Jun Huang*, and Zhen Yang*. Total Synthesis of Penicibilaenes Enabled by a Tandem Double Conia-ene Type Reaction. J. Am. Chem. Soc. 2024, 146, 4363-4368.
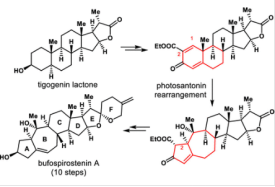
8. Jun Huang*, Tingting Cao, Zhongchao Zhang, and Zhen Yang*. Semisynthesis of (−)-Bufospirostenin A Enabled by Photosantonin Rearrangement Reaction. J. Am. Chem. Soc. 2022, 144, 2479-2483.
9. Zhongchao Zhang, Sijia Chen, Fu Tang, Kai Guo, Xin-Ting Liang, Jun Huang*, Zhen Yang*. Total Synthesis of (+)-Cyclobutastellettolide B. J. Am. Chem. Soc. 2021, 143, 18287-18293.
10. Yongzheng Qu, Zheyuan Wang, Zhongchao Zhang, Wendou Zhang, Jun Huang*, Zhen Yang*. Asymmetric Total Synthesis of (+)-Waihoensene. J. Am. Chem. Soc. 2020, 142: 6511-6515.
11. Tingting Cao, Lei Zhu, Jun Huang*, Zhen Yang*. Palladium-Catalyzed Intramolecular Diarylation of 1,3-Diketone in Total Synthesis of (±)-Spiroaxillarone A. Org. Lett. 2022, 24, 1491-1495.
12. Lijie Zhang, Rongya Wang, Chao Wang, Bingyan Liu, Jinfeng Yang, Zhongchao Zhang, Jun Huang*, Zhen Yang*. Concise Synthesis of 7-Deoxypsammaplysins K and O and 7-Deoxyceratinamide A by 1,3-Dipole Cycloaddition. Org. Lett. 2022, 24, 3786-3791.
13. Jiangqun Cheng, Yuan-He Li, Jun Huang*, Zhen Yang*. Total Syntheses of Vicinal Dichloride Monoterpenes Enabled by AzaBellus-Claisen Rearrangement. Org. Lett. 2021, 23, 8465-8470.
14. Qian Tu, Zheyuan Wang, Zhongchao Zhang, Jun Huang*, Zhen Yang*. Synthetic Strategy for Construction of Highly Congested Tetracyclic Core (6–5–7–4) of Harziane Diterpenoids. Org. Lett. 2021, 23, 4088-4093.
15. Tingting Cao, Lei Zhu, Yu Lan, Jun Huang*, Zhen Yang*. Protecting-Group-Free Total Syntheses of (±)-Norascyronones A and B. Org. Lett. 2020, 7, 2517-2521.
16. Kai Guo, Zhongchao Zhang, Anding Li, Yuanhe Li, Jun Huang*, Zhen Yang*. PhotoredoxCatalyzed Isomerization of Highly Substituted Allylic Alcohols by C-H Bond Activation. Angew. Chem. Int. Ed. 2020, 59, 11660-11668.